Accelerate Research with AI Intelligence
Transform your clinical trials with real-time monitoring and AI-driven insights. Ensure participant compliance while reducing costs by up to 40%.
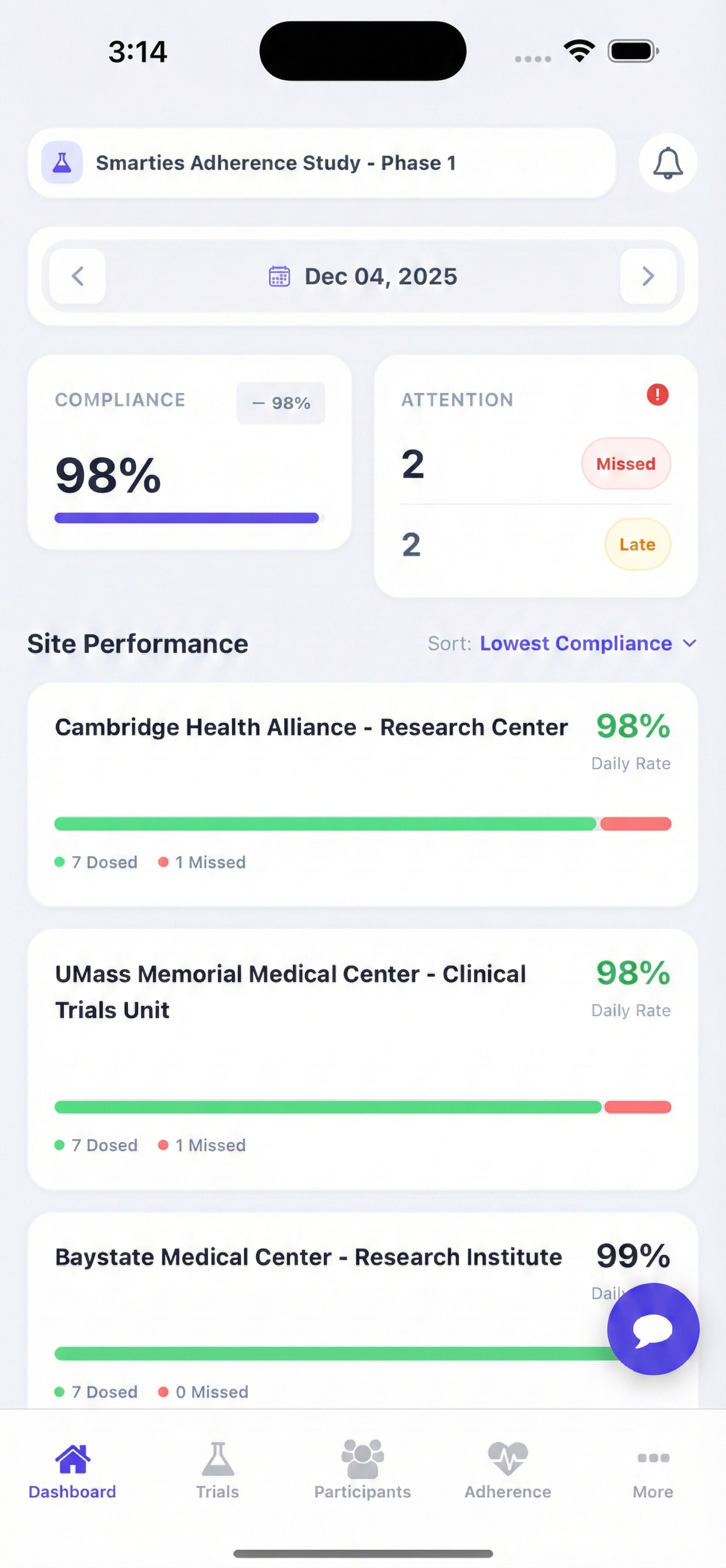
Why Choose Trialsights
Everything you need to run
compliant trials
Leverage AI technology to ensure compliance, reduce risks, and accelerate research outcomes with our comprehensive platform.
Real-time Compliance Monitoring
Advanced AI algorithms track participant adherence and flag potential compliance issues before they impact your trial.
Mobile Participant Engagement
User-friendly mobile app keeps participants connected and engaged throughout the trial process.
Intelligent Analytics Dashboard
Comprehensive insights for labs and sponsors with predictive analytics and compliance trends.
Cost-Effective Solution
Reduce monitoring costs while improving data quality and participant retention.
Enterprise-Grade Security & Compliance
We understand that data security is paramount in clinical research. Our platform is built from the ground up to exceed global regulatory standards.
FDA 21 CFR Part 11 Ready
Designed for full compliance with electronic record and signature requirements, including audit trails and data integrity controls.
HIPAA & GDPR Ready
Built-in privacy controls and architecture designed to protect PHI and assist with your data protection compliance obligations.
SOC 2 Type II Ready
Built with security controls derived from SOC 2 Trust Services Criteria, ensuring robust security, availability, and confidentiality.
End-to-End Encryption
AES-256 encryption for data at rest and TLS 1.3 for data in transit, ensuring maximum information security.
Frequently Asked Questions
Common questions from clinical operations leaders about security, compliance, and implementation.
Is Trialsights compliant with FDA 21 CFR Part 11?
How is participant data secured?
Can Trialsights integrate with our existing EDC system?
What is the typical implementation timeline?
Ready to Transform Your
Clinical Trials?
Join leading research organizations using Trialsights to revolutionize trial compliance monitoring and data integrity.
No credit card required for demo • SOC2 Ready